 
118 There are now 28 type I keratin genes (17 epithelial and 11 hair keratins) and 26 type II keratin genes (20 epithelial and 6 hair keratins). 117 There is even a new consensus nomenclature for mammalian keratins. In the last few years, our knowledge on keratins has tremendously increased, with discovery of many new keratin genes (now numbered at 54 keratin genes) however, most newly discovered are expressed in hair follicles. 108–110,116 This development was crucial for the ease of keratin subtyping that is now indispensable to the surgical pathologist. 110,115 More recently, the problem of keratin subtyping has been expedited by the development of numerous monoclonal keratin-specific antibodies. The original methods for identification of the different keratin types in tissues relied on tedious biochemical methods, chiefly performed by Franke and Moll and their associates. In addition, most low-molecular-weight (LMW) keratins are typically found in all epithelia except squamous epithelium, whereas high-molecular-weight (HMW) keratins are typical of squamous epithelium. 112–114 Within each group, the CKs are numbered consecutively from highest to lowest molecular weight in each group. 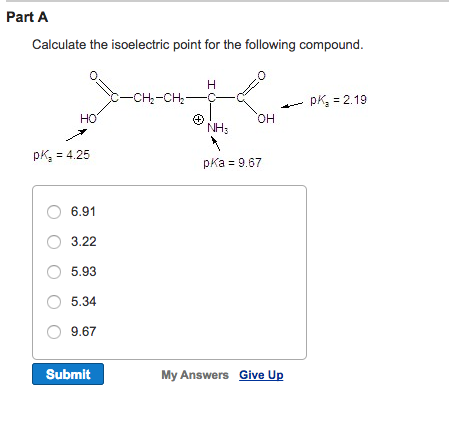
111 The keratins are products of two gene families: most genes for type II keratins are localized on chromosome 12 and the genes for type I keratins are localized on chromosome 17. There are 12 keratins with more acidic isoelectric points that form type I (acidic) keratins and 8 keratins with more basic isoelectric points, the type II (basic-neutral) keratins. The classification and numbering system of the keratins is based on the catalog of Moll and associates. The vast majority of keratins are paired together as acidic and basic types, with rare exception. Keratin filaments are formed by tetrameric heteropolymers of two different keratins, two from type I and two from type II, to maintain cellular electrical neutrality. This family of intermediate filaments is crucial in diagnostic IHC for the identification of carcinomatous differentiation and for identification of specific carcinoma subtypes. 107–109 The polypeptides, numbered 1 through 20, comprise the type II (basic) keratins and the type I (acidic) keratins ( Table 8.2). The soft epithelial keratin intermediate filaments comprise approximately 20 different keratin polypeptides. Dabbs MD, in Diagnostic Immunohistochemistry, 2019 Step Two: The Cytokeratins-An Overview Either peroxidase or alkaline phosphatase may be used as the enzyme label.ĭavid J. Secondary and labeling reagents based on these principles are available commercially that can provide substantial increases in sensitivity and also allow for increased dilution of expensive primary antibodies. 3.īecause the enzyme is directly conjugated to streptavidin in the B-SA system, it is a highly stable reagent that can be diluted and stored for long periods in a ready-to-use (RTU) form. The isoelectric point of streptavidin is close to neutrality, whereas avidin has an isoelectric point of 10 thus, streptavidin conjugates exhibit less nonspecific electrostatic binding than avidin conjugates. Streptavidin contains no carbohydrates, which if present can bind nonspecifically to lectin-like substances found in normal tissue from kidney, liver, brain, and mast cells. The use of streptavidin is preferred to avidin for several reasons: 1. Streptavidin, a tetrameric 60-kDa avidin analog isolated from the bacterium Streptomyces avidinii, is capable of binding biotin with a very high affinity-approximately 10 times higher than that of most antibodies for their antigens. 
The B-SA method overcomes several of the problems associated with the ABC systems by substituting streptavidin for avidin and directly conjugating the streptavidin to the enzyme molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
